Liver Condition Guide
Chronic Hepatitis B (HBV)
A manageable chronic condition — with the right monitoring and treatment.
Chronic Hepatitis B affects approximately 296 million people worldwide and is the leading cause of liver cancer globally. While there is no complete cure yet, modern antiviral therapy can suppress the virus to undetectable levels, dramatically reducing the risk of cirrhosis and liver cancer. Consistent monitoring of viral load, liver function, and cancer screening is the foundation of successful HBV management.

Understanding Chronic Hepatitis B
Hepatitis B is caused by the HBV virus infecting liver cells. In adults, 90–95% clear the virus naturally. But for those infected in infancy or childhood (the majority of chronic cases), the virus persists for life. Chronic HBV cycles through immune phases — from immune-tolerant to immune-active to inactive carrier to reactivation. Understanding your phase is key to knowing when treatment is needed and what to monitor.
Phases of Chronic Hepatitis B
HBV progresses through immunological phases that determine treatment decisions: **Immune Tolerant Phase** — High viral load, normal ALT, minimal liver damage. Common in young patients infected at birth. Monitoring, not treatment, is typical. **Immune Active Phase (HBeAg-positive)** — Rising ALT, active inflammation as immune system attacks infected cells. This is when antiviral treatment is usually started. **Inactive Carrier Phase** — HBeAg seroconversion has occurred, HBV DNA low (<2000 IU/mL), ALT normal. Generally good prognosis but requires ongoing monitoring. **HBeAg-Negative Active Phase** — HBeAg is negative but HBV DNA and ALT are elevated. This phase requires treatment to prevent progression. **Occult HBV** — HBsAg negative but HBV DNA detectable at low levels. Important in immunosuppressed patients.
Key Labs to Track
**HBV DNA (Viral Load)** — The most important monitoring test. Target: undetectable (<20 IU/mL) on antiviral therapy. Checked every 3–6 months. **HBsAg (Surface Antigen)** — Quantitative HBsAg levels predict treatment response and guide duration. HBsAg loss (functional cure) is the ideal but rare outcome. **HBeAg and Anti-HBe** — Seroconversion (HBeAg loss + anti-HBe gain) is a major milestone indicating reduced viral replication. **Anti-HBs (Surface Antibody)** — Protective antibody. Critical post-transplant: levels must stay ≥10 mIU/mL (ideally ≥100) with HBIG prophylaxis. **ALT** — Liver enzyme indicating inflammation. Persistent elevation >2× ULN with high viral load triggers treatment initiation. **AFP (Alpha-Fetoprotein)** — Tumor marker for hepatocellular carcinoma. Checked every 6 months in at-risk patients. **FibroScan / FIB-4** — Non-invasive fibrosis assessment to determine if cirrhosis has developed.
Treatment Overview
**First-line antivirals:** - **Entecavir (Baraclude)** — Potent, high barrier to resistance. Preferred first-line. - **Tenofovir DF (Viread)** — Equally effective, safe in pregnancy. Watch for renal/bone effects. - **Tenofovir AF (Vemlidy)** — Newer formulation with better renal/bone safety profile. **Treatment goals:** 1. Viral suppression: HBV DNA undetectable 2. ALT normalization 3. HBeAg seroconversion (if HBeAg-positive) 4. Functional cure: HBsAg loss (rare but ideal) **Duration:** Typically indefinite for most patients. Stopping antivirals carries risk of severe flares and reactivation — never stop without hepatologist guidance.
HCC Screening
Hepatitis B is unique among liver diseases: it can cause liver cancer even WITHOUT cirrhosis, though cirrhosis dramatically increases the risk. **Who needs screening:** - All HBV patients with cirrhosis - Asian males over 40 and Asian females over 50 - African and African American patients over 20 - Anyone with a family history of HCC **Screening protocol:** Liver ultrasound + AFP every 6 months. LiverTracker tracks your AFP trends and reminds you when screening is due.
Post-Transplant HBV Management
HBV patients who receive liver transplants require lifelong prophylaxis to prevent graft reinfection: **HBIG (Hepatitis B Immunoglobulin)** — Administered IV or IM to maintain anti-HBs titers. Frequency varies: typically monthly early post-transplant, then adjusted based on titers. **Antiviral therapy** — Continued indefinitely post-transplant (entecavir or tenofovir). **Anti-HBs monitoring** — Target ≥100 mIU/mL in early post-transplant, ≥10 mIU/mL long-term. LiverTracker tracks these titers and alerts when HBIG may be needed. **HBV recurrence** — With modern prophylaxis, recurrence rate is <5%. HBsAg reappearance or detectable HBV DNA indicates recurrence and requires urgent intervention.
How LiverTracker Helps
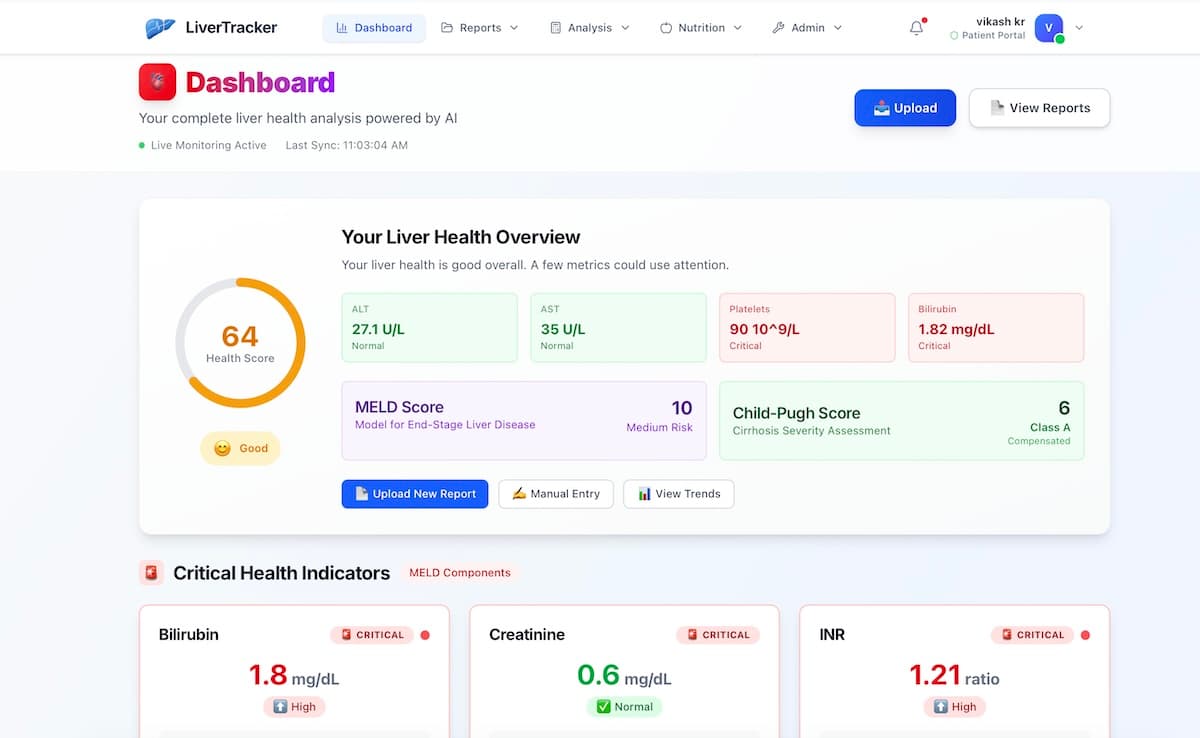
LiverTracker provides comprehensive HBV monitoring: - **Viral load tracking** — HBV DNA trends over time with breakthrough detection - **Antiviral adherence** — Daily medication tracking and reminders - **HCC screening reminders** — Never miss an AFP or ultrasound - **Liver function trends** — ALT, AST, bilirubin, albumin, platelets - **Fibrosis assessment** — FIB-4, APRI, and FibroScan tracking - **Post-transplant monitoring** — Anti-HBs titers, HBIG tracking, immunosuppression management - **AI health assistant** — HBV-aware chat that understands your phase, medications, and lab trends - **Food safety guidance** — Knows to flag aflatoxin-risk foods and alcohol
Key Labs to Track
HBV DNA (Viral Load)
Measures active viral replication. Target: undetectable (<20 IU/mL). The most important single test for HBV management.
HBsAg (Quantitative)
Tracks viral antigen levels. Declining HBsAg correlates with treatment response. Loss of HBsAg = functional cure.
HBeAg / Anti-HBe
HBeAg seroconversion is a major treatment milestone indicating reduced viral replication.
Anti-HBs (Surface Antibody)
Critical post-transplant: must stay ≥10 mIU/mL to prevent graft reinfection. Guides HBIG dosing.
ALT (Alanine Aminotransferase)
Primary liver inflammation marker. Persistent elevation >2× ULN with high viral load triggers treatment.
AFP (Alpha-Fetoprotein)
Tumor marker for liver cancer screening. Checked every 6 months in at-risk patients.
FIB-4 / FibroScan
Non-invasive fibrosis assessment. Determines if cirrhosis has developed and guides transplant evaluation.
Platelets
Low platelets indicate portal hypertension and cirrhosis. Used in FIB-4 calculation.
How LiverTracker Helps
Tracks HBV DNA viral load trends over time with breakthrough detection alerts
Monitors antiviral therapy adherence and medication schedules
Sends HCC screening reminders (AFP + ultrasound every 6 months)
Visualizes liver function trends — ALT, AST, bilirubin, albumin
Calculates fibrosis scores (FIB-4, APRI) and tracks progression
Post-transplant: monitors anti-HBs titers, immunosuppression levels, and graft function
AI health assistant trained on HBV guidelines — answers questions about your specific situation
Flags aflatoxin-risk foods and drug interactions in the food scanner
Shares reports with your hepatologist through secure doctor sharing links
See Your Data Come to Life
Upload your lab reports and get instant AI-powered insights, trend charts, and health scores.
Frequently Asked Questions
Can Hepatitis B be cured?
A complete cure (eliminating all HBV DNA including cccDNA) is not yet possible. However, a 'functional cure' — defined as HBsAg loss — can be achieved in about 1–3% of patients per year on treatment. Most patients achieve viral suppression (undetectable HBV DNA), which effectively prevents liver damage progression.
How long do I need to take antiviral medication?
For most patients, antiviral therapy is indefinite. Stopping antivirals — even after years of viral suppression — can lead to dangerous viral reactivation and liver flares. Some patients with HBeAg seroconversion and sustained suppression may be candidates for treatment withdrawal under very close monitoring, but this must be discussed with your hepatologist.
Can I spread Hepatitis B to my family?
HBV spreads through blood, sexual contact, and from mother to child during birth. It does NOT spread through casual contact, sharing food, coughing, or hugging. All household members and sexual partners should be tested and vaccinated if not immune. The HBV vaccine is 95% effective.
Why do I need liver cancer screening if I don't have cirrhosis?
Unlike most other liver diseases, HBV can cause liver cancer (HCC) even without cirrhosis because the virus integrates into liver cell DNA. Your risk depends on factors like age, gender, ethnicity, family history, and viral load. Your hepatologist will determine your screening schedule.
What does my HBV DNA viral load number mean?
HBV DNA measures active viral replication in IU/mL. Undetectable (<20 IU/mL) is the goal on treatment. 2,000 IU/mL is the threshold that often triggers treatment decisions. Above 20,000 IU/mL indicates very active replication. LiverTracker tracks your viral load trend so you can see your progress over time.
Is it safe to have children with Hepatitis B?
Yes, with proper management. If the mother is HBV-positive, tenofovir is preferred during pregnancy (especially in the third trimester) to reduce vertical transmission. The newborn should receive HBIG + first HBV vaccine dose within 12 hours of birth. With these measures, transmission risk drops to <5%.
Related Conditions
Take Control of Your Liver Health
Upload your lab reports, track your progress, and get AI-powered insights — all for free.
Get Started FreeMedical Disclaimer: This page is for educational purposes only and does not constitute medical advice. Always consult your hepatologist or primary care physician for diagnosis and treatment decisions.